
26 March 2026
ubispoke®: Chemistry-Centric Manufacturing of Advanced Intermediates
The specialty chemicals industry is facing increasing pressure to deliver complex molecules reliably, cost-effectively, and at scale, while managing regulatory expectations and global supply chain risk. In response, manufacturing models are evolving beyond traditional site-centric approaches. With ubispoke®, learning from 50 years of market leadership we have developed a modern manufacturing model designed to combine technical control, flexibility, and global reach - positioning ubispoke® as a technically engaged manufacturing partner.
A Manufacturing Footprint Designed Around Chemistry
At ubispoke®, manufacturing programmes are bespoke, built around the specific needs of the chemistry and the the customer, rather than the constraints of a single site.
This approach provides access to a broad array of chemical capabilities and technologies, enabling the execution of projects ranging from early development through to commercial scale.
Each programme operates within a controlled manufacturing model, selected to align with chemistry, scale, regulatory pathway, and long-term supply strategy. This ensures consistency of execution while maintaining flexibility to adapt as project requirements evolve.
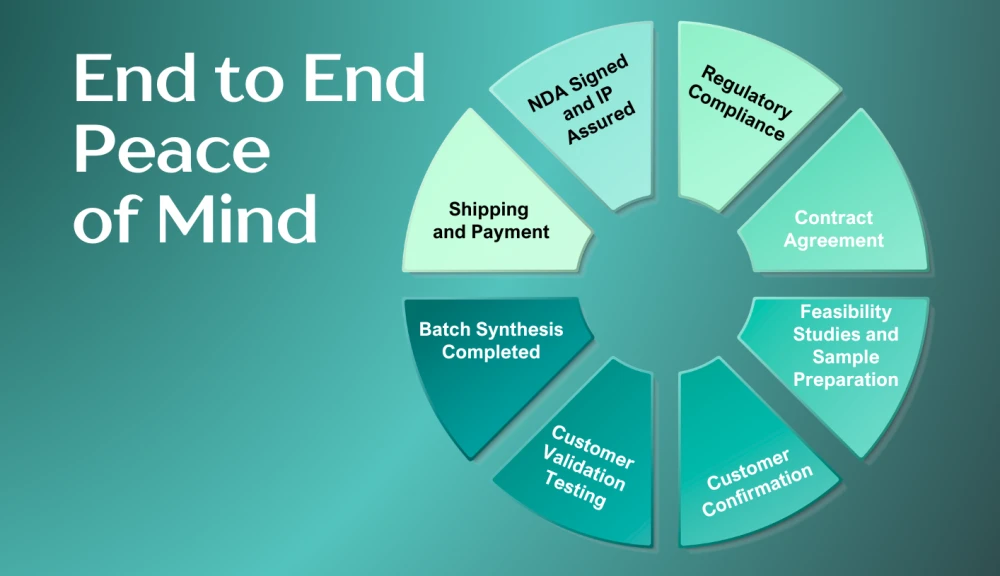
Technical Ownership and Process Equity
Manufacturing control at ubispoke® is driven by deep technical ownership. ubispoke® teams are directly involved in route selection, process development, optimisation, and scale-up, maintaining process leadership throughout the product lifecycle. This ensures continuity of knowledge, robust change control, and effective protection of intellectual property.
Technical change, whether driven by optimisation, raw material availability, or scale requirements, is governed through a structured framework that defines decision-making, documentation, and regulatory oversight. As a result, changes are implemented predictably and without disruption, preserving regulatory alignment and continuity of supply from variable chosen geographic locations.
End‑to‑End Programme Management: A True Manufacturing Partner
ubispoke® operates as a one‑stop manufacturing partner, integrating project management across technical execution, regulatory oversight, manufacturing deployment, logistics, and warehousing from initial feasibility through to commercial supply. This single, coordinated structure creates clear accountability and enables decisions to be made with full visibility of downstream impact.
By combining strong global logistical expertise with customer relevant warehousing, ubispoke® actively controls material flow and lead times, allowing manufacturing output to be adjusted in line with real customer demand rather than fixed production schedules.
Flexibility, Scale, and Supply Resilience
Flexibility in scale is at the heart of the ubispoke® manufacturing strategy. Programmes are scaled efficiently as demand evolves, without fundamental changes to process or supply strategy. Geographic options and multiple sourcing strategies built with redundancy are core to programme design, enhancing resilience against regional disruption and supply chain volatility.
Customers no longer have to rely on closed or single-location supply chains, they can chose ubispoke® to deliver trusted, adaptable manufacturing strategies that support their long-term commercial success.
Cost Effectiveness Beyond Price per Kilogram
Cost effectiveness at ubispoke® is considered holistically. While competitive unit pricing is important, equal emphasis is placed on development efficiency, robustness of process, reliability of supply, and lifecycle cost.
By aligning chemistry, scale, and manufacturing strategy from the outset, ubispoke® helps customers avoid costly rework, delays, and supply interruptions. The result is a manufacturing solution that delivers value well beyond headline pricing.

Experience That Builds Confidence
As ubispoke® continues to grow, its team brings substantial experience across specialty chemicals and regulated manufacturing environments. This experience is demonstrated through successful project management involving novel and complex chemistries, process optimisation, and controlled technical change.
Trust is built through delivery. ubispoke® emphasises transparency, technical depth, and accountability, enabling long-term partnerships, more than transactional engagements.
A Partner for the Next Generation of Manufacturing
As specialty chemical portfolios become more specialised and supply chains more complex, manufacturers must balance control with flexibility. ubispoke® is an execution model designed to meet this challenge, combining technical expertise, global reach, and disciplined governance into a single manufacturing offering.
For customers seeking a technically engaged, reliable, and adaptable manufacturing partner, ubispoke® represents a modern tried and tested approach to delivering chemistry at scale.
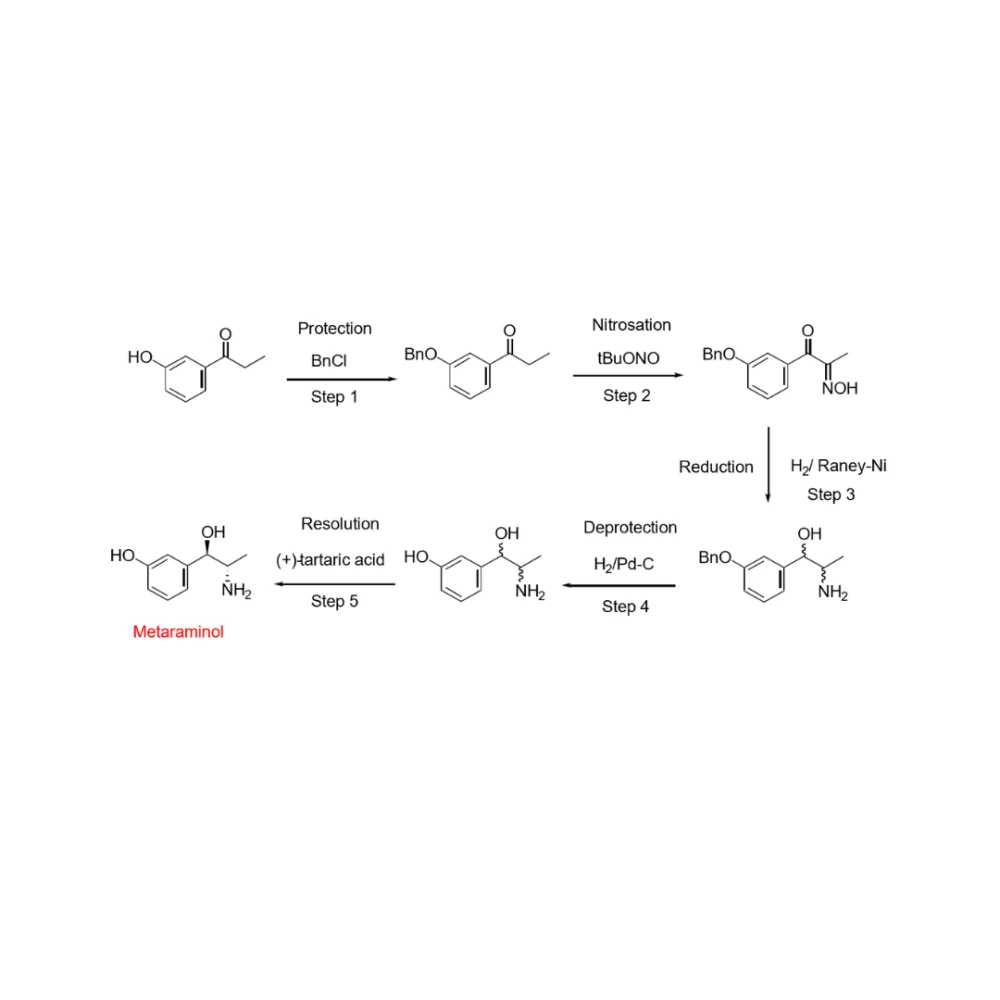
Redefining Amino Alcohol Synthesis: Greener, Safer, and More Efficient Chemistry
As an example of effective project management and process development, the synthesis of amino alcohols has traditionally relied on conventional forward synthetic routes. These approaches often involve multiple steps, including protecting‑group chemistry, nitrosation, hydrogenation, and final resolution, resulting in lengthy processes that place significant demands on plant capacity, personnel, and quality and regulatory oversight.
High material losses were common, with a substantial proportion of input material diverted to waste, increasing both environmental impact and overall cost. In addition, the use of nitrosation chemistry introduced a potential risk of nitrosamine formation - a safety and compliance concern that many organisations now seek to eliminate from their manufacturing strategies.
More recently, this route has been transformed into a markedly greener, safer, and more efficient process. The key initial step now uses biotransformation with the carboligase enzyme ECAHAS-1, sodium pyruvate as the substrate, and 3-hydroxy benzaldehyde as starting material. This enzymatic transformation proceeds under mild, low-temperature conditions, delivering 95% conversion, 95% isolated yield, and an enantiomeric excess of 99% for the desired (R)-isomer.
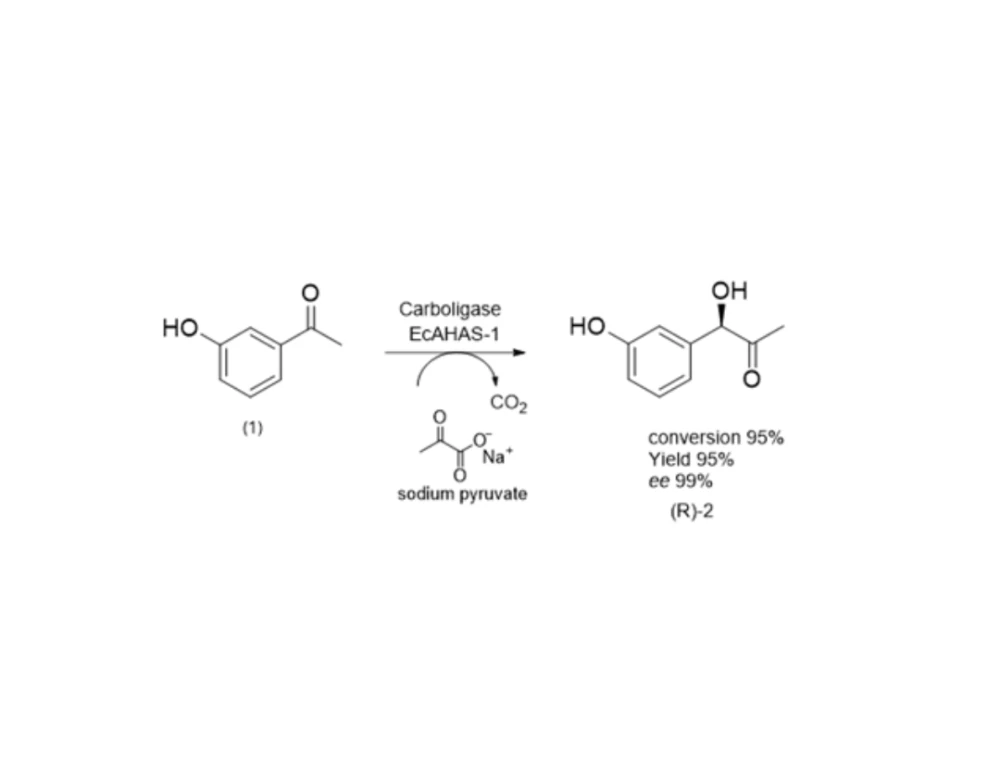
This elegant approach eliminates the need for protecting groups and removes the nitrosation risk entirely, while introducing complete stereochemical control in a single step. By significantly reducing waste, lowering energy usage, and improving overall yield, the process frees up plant capacity, reduces regulatory and quality burden, and provides a more sustainable and industrially attractive route to the API. In addition to enhancing operational efficiency, this greener process aligns with modern pharmaceutical priorities of safety, sustainability, and cost-effectiveness.
More broadly, this example reflects the ubispoke® approach to manufacturing execution - applying technical ownership, disciplined change control, and flexible deployment to deliver better outcomes across the product lifecycle. It demonstrates how a technically engaged manufacturing partner can drive safer, more sustainable, and commercially robust solutions, without compromising control or regulatory integrity.

